The University announced today a gift of $131 million from Hansjörg Wyss, M.B.A. ’65, to support the operations of the Wyss Institute for Biologically Inspired Engineering. This is the third such gift that the Swiss-born entrepreneur has made to the institute, which seeks to bridge the gap between academia and industry by drawing inspiration from nature’s design principles to solve some of the world’s most complex challenges in healthcare and the environment, and to commercialize those solutions to maximize their impact.
Wyss’s support for the institute’s unique model—interdisciplinary collaboration among scientists and engineers from disparate fields working together with expert staff who have industrial experience—has led to a remarkable track record of innovation and success in translational work in numerous scientific fields, especially in biomedicine and biomedical engineering. Immunotherapies for cancer, organs-on-chips that enable drug testing on living human tissue, and clinic-ready molecular diagnostic tools that work to rapidly detect diseases are among the life-altering results. “In the last decade, the Wyss Institute has made breakthrough after breakthrough to improve medicine and to apply the latest science to the betterment of peoples’ lives,” said Wyss. “I am happy to continue my support for the Wyss Institute and Harvard and look forward to seeing what the Institute discovers and creates in the years ahead.”
The Donor
Wyss was CEO of Synthes, a biomedical research and device company that pioneered treatments such as plating and screwing (rather than casting) after traumatic bone breaks, which sped patient healing and recovery. “The company started with fixation of the legs and arms, but soon expanded into maxillofacial applications, and treatments for the feet, hands, and spine,” recalls Robert Bland ’62, a friend of Wyss who served on the Synthes board, and is now a member of the board of the Wyss Institute. “He always said that if [a treatment] benefited patients, then it would be good for the company.” Wyss held his product managers to high standards, said Bland, and “his understanding of human nature and motivation” and ability to pick good leaders led to success at Synthes and later, at his philanthropic foundations. Wyss sold Synthes to Johnson & Johnson in 2012 for a reported $20 billion.
He is well known not only for his engagement with and support of science and engineering initiatives, but also for his dedication to land conservation (often with a social-justice component) and improved fisheries-management policies around the globe. Through the Wyss Foundation, he has partnered with organizations such as the Nature Conservancy and Trout Unlimited to creatively protect in perpetuity more than 40 million acres of land and wildlife habitat in the American West, in Canada, Africa, South America, and Europe—more than any other living person. This spring, he was elected to membership in the American Academy of Arts and Sciences in recognition of his achievements in the field of business, corporate, and philanthropic leadership.
In 2009, Wyss donated $125 million (then the largest gift ever made to Harvard), to launch what has become the eponymous enterprise in Boston. He then committed another $125 million in 2013, once the organization’s success had been demonstrated and it had been incorporated as a 501(c)3 nonprofit. Wyss has also given $25 million to Harvard Business School (HBS), to establish the Hansjörg Wyss Endowment for Doctoral Education there, as well as $2.8 million to the Medical School, a portion of which established a chaired professorship there, and $500,000 to the Harvard Art Museums. His giving over many years and across the University amounts to more than $400 million, making him one of the most generous donors in University history, and this third gift to the bioengineering institute further strengthens Harvard’s leadership position in the field.
Recognizing this new gift and the continuing progress of the institute, President Lawrence S. Bacow said, “Hansjörg Wyss has helped to expand what we know and what we can accomplish across a wide range of disciplines. The advances that his generosity has enabled will change the future for countless people. His third gift to support the work of the Wyss Institute will ensure the continued success of our extraordinarily talented faculty and staff, as well as create new opportunities for collaboration and growth. We are deeply grateful for his support.”
“He always said that if [a treatment] benefited patients, then it would be good for the company.”
University provost Alan Garber, an economist and physician who leads Harvard’s life-sciences initiatives and chairs the Wyss board of trustees, added that the institute has advanced the field of bioengineering significantly. “From developing singular insights and cutting edge approaches, to creating bioinspired materials and feats of engineering, the Wyss Institute had and will continue to have a powerful impact,” Garber said. “Biologically inspired engineering was an unconventional approach when the Wyss Institute was created in 2009, but it is now recognized as a powerful approach to solving a wide range of problems,” Garber elaborated in an email. “As Susan Hockfield, president emerita and professor of neuroscience at MIT, argues in her recent book The Age of Living Machines: How Biology Will Build the Next Technology Revolution, discoveries in this broad area have the potential to be as transformative as the twentieth century’s digital revolution. Some of the technologies that have come from the Wyss Institute are simply astounding, such as…3-D bioprinting of living tissue, and versatile ambulatory microrobots. The Wyss Institute was created to move the scientific advances of bioengineering faculty at Harvard and collaborating institutions beyond academic labs, developing real-world products that treat and diagnose human disease. Endeavors to translate research advances in this way have a long history; the Wyss Institute has demonstrated that we can do so quickly and successfully.”
The Institute’s Origins: “You Have to Continue”
The origins of the institute trace to a three-day visit Wyss made to Harvard in 2005, during which he met with faculty and saw “great research,” he said in a recent interview. “But it was all in a silo.” Wyss remembered meeting “brilliant professors” who attended the same conferences and did the same types of studies as some of their colleagues, but had never worked with them. During lunch on the last day, when a discussion arose about molecules that had been discovered within meteorites, Wyss assumed that the astronomers who’d made the discovery would immediately want to work with molecular biologists to study what they’d found. But when he articulated the idea, he recalled, “The group…looked at me as if I was from the moon.”
People who have known Wyss for a long time describe him variously as competitive, driven, and intellectually curious; creative; and entrepreneurial. Unsurprisingly then, he approached HBS professor Howard Stevenson (now emeritus), who was then University vice provost, as well as a friend and HBS classmate, and said, “Howard, we’ve got to do something.”
Wyss approached HBS professor Howard Stevenson (now emeritus), who was then University vice provost, and said, “Howard, we’ve got to do something.”
Harvard at the time was exploring ways to organize its bioengineering research efforts across the entire University and its affiliated hospitals, guided by a committee co-chaired by the Medical School’s Folkman professor of vascular biology Don Ingber, an affiliate of Children’s Hospital Boston, and Pinkas Family professor of bioengineering David Mooney. “It was fairly clear to me,” Stevenson recalled in an interview, that “this might excite Hansjörg’s interest, because he’d been very innovative in his corporate life with Synthes. And if you think about what he was doing there, it was, ‘How do you marry materials with biology?’”
Wyss said he wanted to put physicians, physicists, mathematicians, mechanical engineers—every pertinent discipline—together to work on various projects. He met with then Harvard provost Steven Hyman and reached an agreement: he would give a large gift, and the University would support the effort financially, too. (Wyss emphasized that there would be no money for construction of a building; the funds were purely for science and research.)
In a frenzied six months, with input and assistance from Hyman’s office, Mooney and Ingber, who is also professor of bioengineering at the Harvard Paulson School of Engineering and Applied Sciences (SEAS), drew up a business plan that encompassed both the University and teaching hospitals, and several other Boston-area research institutions, to create an affiliated organization like the already existing Broad Institute (which focuses on genomics and and biomedicine). Because Mooney preferred to continue focusing on research, Ingber took on the role of founding director. The organization’s success, he said in a recent interview, would be measured in terms of “academic objectives, including its ability to attract great people and international recognition, as well as its intellectual-property portfolio, licensing agreements, startups, corporate alliances, and having product in the pipeline in five years. I had this money and I had to figure out, How do we get this up and running? I had a five-year timeline, so this would die in five years, with no promises of additional funding.”
“I had this money and I had to figure out, How do we get this up and running? I had a five-year timeline, so this would die in five years, with no promises of additional funding.”
Five years later, what had begun as an experiment had clearly proved itself. “When you see the research that has been done in the first five years, the intellectual property, the papers published in top scientific magazines,…the influence it had on teaching at Harvard, the influence it had on collaborative research throughout [SEAS] and the Faculty of Arts and Sciences—you have to continue,” said Wyss, speaking from Paris. Having just looked at some great paintings, he added, “When Cezanne created one of his landscapes, he had to go and paint another one. He could not stop. So, when you create the Wyss and then think about the second gift, you almost have to do it.”
The Science: “A Startup on Steroids”
Despite the large sums involved (to date, $381 million from Wyss and $187 million from the University), the institute’s beginnings in 2009 were humble, Ingber recalled. Its first employee, Mary Tolikas, who quit her job at a startup and joined the organization as operations director, and Ayis Antoniou, the second employee and now Wyss administrative director, at first held meetings in cars parked strategically to tap the local wireless network, trying to launch a completely new kind of translational research organization that would draw engineering inspiration from nature.
Within three years, though, there were 300 full-time staff members. (There are now about 375 staff members, housed in about 100,000 square feet of leased space, mainly in the Longwood Medical area, with a small presence in Cambridge.) “It was like a startup on steroids” said Ingber, “and at the time we couldn’t believe we’d done it.” Since its launch, the Wyss has produced 2,085 publications, with one paper in Science or Nature per month on average (the holy grail for scientists publishing their discoveries); filed for 2,623 patents—and been issued 392; launched 29 startups; and entered 53 agreements to license technology. Since 2009, 23.8 percent of Harvard’s patent applications have been linked to Wyss research.

An organ-on-a-chip
Courtesy of the Wyss Institute at Harvard University
The institute’s scientific program is organized around constantly evolving “platforms” and initiatives (see below), each aiming to foster technologies that are developed and de-risked through the institute’s unique validation and project advancement program, which has launched numerous startup companies and entered industrial licensing agreements to commercialize technologies developed at the Wyss. Current areas of research include:
- soft, wearable robots that increase the user’s balance, endurance and strength, either in ill-health or in environments that place extreme demands on the human body;
- adaptive materials that are responsive and self-optimizing, such as coatings that prevent barnacles from sticking to the hulls of ships, thereby producing huge energy savings;
- biomimetic microsystems, such as organ-on-a-chip microfluidic cell culture devices that recapitulate the complex functions and disease states of living human organs—useful for drug testing and personalized medicine;
- materials that can modulate immune responses to treat cancer and infections, for example;
- engineered, living, cellular devices programmed to detect pathogenic viruses, or reduce off-target drug delivery;
- programmable swarms of molecular robots that can aid in sequencing, imaging, and drug delivery;
- synthetic biology, for editing genes and proteins in applications ranging from healthcare and chemical manufacturing to information storage; and functional, vascularized 3-D organ replacements for implantation in the human body.
An early example of the push for clinical applications involved the use of stochastic resonance (a form of low-level noisy vibration) in two completely different—and independent—applications. In one, Dr. David Paydarfar, then a professor at the University of Massachusetts Medical School and an associate faculty member at the Wyss Institute, used such vibration to help address breathing difficulties, called apnea of prematurity, in preterm infants. Because these infants’ respiratory systems are not fully developed, physicians have traditionally treated them with caffeine. Paydarfar found that a stochastically vibrating mattress, stimulating at a very low level, might encourage the infants’ pacemaker neurons to maintain normal breathing function. The Wyss Institute also worked with Paydarfar to develop algorithms which could predict in advance when an apnea event might occur, so the mattress could provide stimulus when needed. The mattress was tested in the neonatal intensive care units at UMass Memorial Hospital as well as at Beth Israel Deaconess Hospital. The technology has been licensed to a company which will pursue a variety of infant health applications.
Stochastic resonance technology also shows promise for improving balance, possibly helping to prevent falls in the elderly, and address other age, injury, and disease-related conditions. James Collins, then a professor at Boston University (and now a professor of bioengineering at MIT, and a core faculty member of the institute), independently developed a vibrating insole that enhances the wearer’s balance. “When you touch a surface,” explained administrative director Ayis Antoniou in an interview, “you either feel it or you don’t feel it. With age, that sensitivity goes down—it takes more pressure and stimulus to feel the same touch.” Stimulation via stochastic resonance, which can’t be consciously detected, has been demonstrated to increase sensitivity to touch, pressure, and joint position.
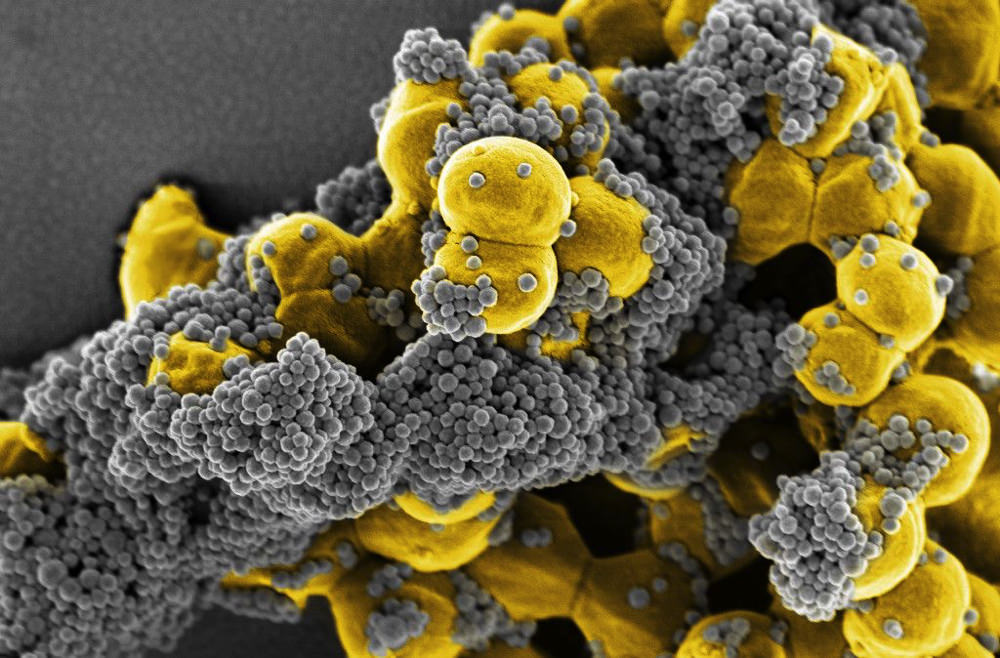
Proteins engineered to capture pathogens to prevent sepsis
Courtesy of the Wyss Institute at Harvard University
The Wyss’s dialysis-like therapy for treating sepsis offers an example of its collaborative production model. Michael Super, a lead senior staff scientist on the institute’s Advanced Technology Team (ATT; see below), was co-principal investigator with Ingber on the project funded by the Defense Advanced Research Projects Agency (DARPA); he took a protein from the innate immune system, and adapted it so that it could be used to filter a patient’s blood and remove the dangerous pathogen products that cause sepsis. “Our aim is to cleanse the blood of toxins and save lives,” Super explained. “We assembled a multi-talented team of 36 with expertise in microbiology, protein engineering, microfluidics, and small and large animals sepsis models, as well as staff to manage bio safety, compliance and grants: the kind of group that wouldn’t typically self-assemble in an academic lab.” Faculty members, staff scientists, postdoctoral fellows, doctoral students, research assistants and administrative staff all worked together on the common goal.
Super’s brief was to “design a system to clear multiple different pathogens, both for biodefense and civilian medicine.” Each year, DARPA, raised the bar and increased the number of pathogens. The protein group designed more than 64 proteins, the microbiology group assembled a large number of clinical pathogens to test, the microfluidics team performed in vitro studies, while the animal teams tested them in vivo. “By the end of the four-year grant,” Super said, “we had shown that our system could bind more than 110 different pathogen species including 15 of the 16 major pathogens that cause sepsis.” The technology has been licensed to a startup company.
“By the end of the four-year grant,” says Super, “we had shown that our system could bind more than 110 different pathogen species including 15 of the 16 major pathogens that cause sepsis.”
The Model: “You Just Get Out of Their Way”
The institute’s technology-translation model has proven extremely effective. “We basically created a structure that enables complex, interdisciplinary, translation-focused science to happen really quickly,” Ingber explained. That matters because “We have a large number of [Defense Department-affiliated] major grants that represent tens of millions of dollars,” and often require rapid response and delivery. (Historically, about half of the institute’s $75-million to $80-million annual operating budget has come from grants; another 30 percent from the Wyss gifts; and the final 20 percent from the University.)
The institute’s own administrative structures, from financial accounting, to management, to grant submissions, institutional review boards, and animal studies, have been set up for efficiency in bringing technological innovations to market. “You can't do disruptive things easily, working within existing structures,” Ingber said. “We hit a lot of pushback in the beginning.”
Of the 375 people who work at the Wyss, 150 to 170 are full-time staff , and the other 200-plus include 18 core faculty, 16 associate faculty, and postdoctoral fellows and students. The exact numbers change frequently, because when a project results in a startup, members of the project team often leave to join the new company. When Don Ingber’s organs-on-chips led to the launch of a company called Emulate, for example, staff scientists, engineers, entrepreneurs, technicians, and postdoctoral fellows left overnight—about 18 people in all. That creates staffing challenges, admitted administrative director Antoniou, but it also creates opportunities to attract new people whose expertise is better suited to future projects.
But perhaps most significant in the Wyss’s efforts to “harness the intellectual power of the Boston region,” as Antoniou put it, was recognizing that “one of the barriers to close collaboration has to do with how you manage intellectual property and share revenues. Every time people try to collaborate beyond the classic free flow of academic collaborations, their home institutions have to put agreements in place”—which “takes forever. And so we decided that it made sense to create an agreement that will govern our relationship with other institutions in the area.” Revenues from intellectual property created at the Wyss are therefore shared according to a uniform governing agreement with its institutional partners, including Children’s Hospital, Beth Israel Deaconess Medical Center, Massachusetts General Hospital, Brigham and Women’s Hospital, Dana-Farber Cancer Institute, Spaulding, Boston University, MIT, Tufts, UMass Medical School, and Charité Hospital in Germany and the University of Zurich. Anytime the agreement is changed, everyone gets the same benefit. And the institute’s board is made up of the donor and his representatives, as well as representatives from these institutions and senior Harvard leadership, including the deans of Harvard’s medical and engineering schools. Venture capitalists have recently joined the board for the first time.
One example of a soft robot developed at the Wyss Institute
Courtesy of the Wyss Institute at Harvard University
Its organization sets it apart from, say, a Harvard academic department or school. In effect, it is a stand-alone research institute, engaging faculty members, students, and many others, embedded within the University, but spanning across multiple disciplines, with different operating methods, and ways of engaging personnel. An analogy might be the Broad Institute, launched as an MIT-Harvard collaboration, but now a stand-alone genomics research center, involving faculty members from the parent institutions and elsewhere, and hundreds of other personnel as well.
Among the institute’s research innovations are its “Enabling Technology Platforms,” which involve finding a visionary in a given area of inquiry, and giving that individual the funds to develop new capabilities that will advance the entire field: yielding not one breakthrough, but technological capabilities. That funding, in fact, requires recipients to enable other faculty members and bring in other people to collaborate. Among them, said Ingber, have been Winthrop professor of genetics George Church, who “headed Synthetic Biology, with a major focus on genome engineering. Dave Mooney started out on Nanomaterials and I headed Biomimetic Microsystems, (which led to organs on chips)”; Berylson professor of materials science and professor of chemistry and chemical biology Joanna Aizenberg, co-director of the Kavli Institute for Bionano Science and Technology, headed Adaptive Architecture and Robotics. “A year later we separated out bio-inspired robotics and brought in Rob Wood,” Charles River professor of engineering and applied sciences, “who is the developer of the ‘Robobee’ flying insect robot, and [Kavli professor of computer science] Radhika Nagpal,” an expert in algorithmic control of robot swarms, “to head that.” James Collins, then a professor at B.U., headed up Anticipatory Medical Devices.” Each of these professors became part of the Wyss Institute’s core faculty, a designation that is reviewed every three years to ensure alignment with the Institute’s translational goals.
But perhaps the most novel of the institute’s innovations, Ingber asserts, was “bringing engineers into the middle of [the Longwood Medical Area], the greatest medical area in the world, and to recruit staff from industry.” These 35 to 40 people, who have decades of experience in product development at existing or former companies such as Pfizer, AstraZeneca US, DuPont and Dow Chemical, Pratt and Whitney (a unit of United Technologies), Google, and iRobot make up the institute’s Advanced Technology Team (ATT). “Because they sit in open spaces,” Ingber explains, “when somebody says, ‘I need something for a knee joint with these lubricant or material properties,’ someone else might say, ‘Well, we used something that had those properties as an airplane-engine insulation at Pratt and Whitney.’ You don't see that at companies because they're usually focused on their expertise,” he pointed out, “and you don't ever see that in academia.”
Pairing faculty members with co-lead investigators from the ATT who have budget and management experience has created an effective synergy, Ingber continued. Because ATT staff can be deployed flexibly as needed across the organization, he said, they essentially “form a matrix across the whole institute.”
The Wyss has also pioneered what might be called self-assembling interdisciplinary research. Instead of erecting buildings and placing experts from different disciplines who have “no reason to work together” in proximity to one another, “You identify problems that are so exciting, and so difficult, and you get the best people who want to solve them, but who can't do it on their own” said Ingber. “And then, when you bring other scientists and engineers that have the right expertise to complement them nearby, you just get out of their way, and it happens.” Because faculty members commonly operate like “independent entrepreneurs, who have their own cultures,” he continued, “we created a new culture: we let our faculty keep their own labs,” but move some portion of it”—generally their most entrepreneurial postdoctoral fellows, research assistants, and graduate students—to the Wyss.
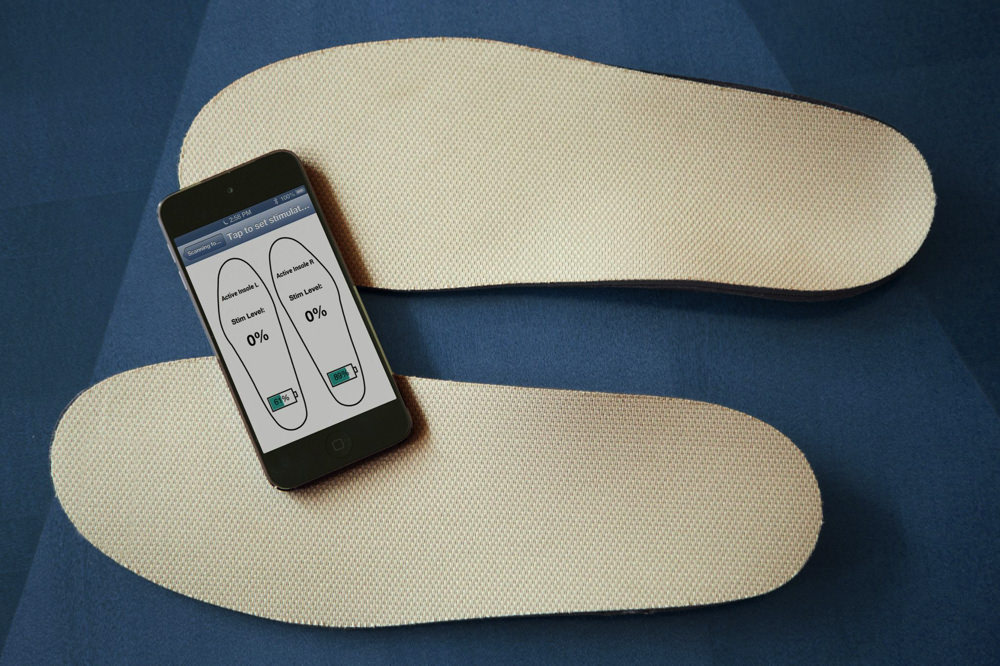
Vibrating insoles can improve balance and agility.
Courtesy of the Wyss Institute at Harvard University
And the Wyss also has the resources and knowhow to bring faculty members’ ideas to the point where they have been de-risked enough so that industry partners are willing to license them. A full-time in-house attorney helps identify and protect intellectual property early in the process. Members of the ATT refine the technology and assess its potential for commercialization. The vibrating insoles are just one example. “The footwear industry really expects projects to be quite far along before they take them,” explained Jim Niemi, a lead senior staff engineer on the ATT with both engineering and business experience. “We had an academic prototype that was interesting, and some very exciting clinical results. But whenever we showed it to a footwear company, they would say, ‘That is interesting, but it's not done yet.’”
“We got that feedback from eight to 10 different well-known, reputable footwear companies,” he continued. They wanted the insole to be less expensive (the Wyss team reduced the costs 90 percent) as well as thinner and lighter, and they wanted evidence that it would help athletes in their prime, not just the elderly or those with sensory-related loss. “We want LeBron James to wear it,” one executive told Niemi. The group then engineered a thinner insole, and were able to show that it improved agility in young people, Niemi said, so “we ended up having a technology with a broader application.”
In Prospect: “A Marvelous Way to Go at Innovation”
Today the Wyss has not only built an international reputation as an innovation engine, said SEAS dean Frank Doyle, but has also played a valuable role in recruiting translationally minded faculty like the school’s Jennifer Lewis, known for her work to develop an artificial kidney. “It’s a marvelous way to go at innovation,” Doyle added, even as he acknowledged that it is not one that would be affordable across an entire academic enterprise that has teaching and other obligations. His own role on the board, Doyle said, is to produce a sustainable, long-term plan for the institute, to maintain the “tremendous opportunity” that it represents.
Can the Wyss model be replicated elsewhere? On that question, there is disagreement. Its location in the heart of one of the most vibrant medical research areas in the world, and Ingber’s innovative leadership, cannot be easily duplicated. But aspects of its culture and organization can certainly be reproduced—and in some cases, have been already.
Howard Stevenson pointed out that many of the practices pioneered by the institute have been adopted widely throughout the University—through its many multidisciplinary centers, at the Harvard Stem Cell Institute, and even within schools, such as the Harvard T. H. Chan School of Public Health, where the research is “no longer in silos.” Expensive core facilities—for imaging, computing, or manufacturing cell lines, for example—are now being run as shared resources for anyone in the University to access. And Harvard’s Office for Technology Development has grown significantly (as many as six members of its staff work at the Wyss).
Said Ingber, “We thought we were going to develop an institute, to create bio-inspired materials and devices. In the end, we developed an entirely new model for disruptive innovation.”










