The light microscope launched modern biology in the seventeenth century, letting scientists view the components of life that exist far beyond the range of unaided human vision. But light travels in waves, and its wavelength prevents researchers from focusing on details below a certain size limit without creating interference. As a result, light microscopes cannot show details that are less than about 200 to 300 nanometers apart. This is fine for viewing a single cell, but look inside it and things blur. Even a cluster of proteins, each a mere three to 10 nanometers in size, appears as an indistinct blob. (A nanometer is a billionth of a meter; a human hair is 80,000 nanometers in width.)
Creative new techniques developed in the past few years, however, have extended the boundaries of what these microscopes can reveal. The latest approach, from the lab of Xiaowei Zhuang, professor of chemistry and chemical biology at Harvard and a Howard Hughes Medical Institute investigator, uses fluorescent molecules that can be switched on and off to create images with more than 10 times the resolution of traditional light microscopes.
Zhuang says that light, despite its limitations, has important advantages for viewing cells. Electron microscopy, though more powerful in resolution, requires that cells be killed and chemically fixed. With electron microscopy, she says, “You’re looking at a still image,” rather than capturing activity in a cell. Light, on the other hand, is noninvasive, so it can be used to image live cells; scientists can use a variety of stains and fluorescent tags that let them view specific proteins or structures of a cell in different colors. Ideally, Zhuang says, “one would want to have a method that combines the merits of both”: the high resolution of electron microscopes paired with the flexibility of optical microscopes.
The new technique, which Zhuang developed with graduate students Michael Rust and Mark Bates, makes use of fluorophores—molecules that absorb light and then fluoresce, emitting light at a different wavelength. Fluorophores have been used in microscopy for many years because they can be attached to specific molecules in cells, allowing scientists to track their location.
Normally, when a sample of fluorophores is exposed to light, they fluoresce in unison. Under an optical microscope, this sea of individual spots cannot be resolved if they are densely packed. Even so, Zhuang says, scientists have known how to determine mathematically the position of a single, isolated fluorophore by calculating the center of the fuzzy spot of light detected by the microscope. The trick was to find a way to turn the lights out on a group of fluorophores, allowing only a few at a time to light up so that the microscope could pick out individual spots.
Several years ago, Zhuang’s group discovered a fluorophore that has an on-off switch—when exposed to red light, it is inactive, but when exposed to green, it has the ability to fluoresce. “This was the additional step that allowed us to control how many molecules are active,” she says. Using this molecule, they can first turn off all the fluorophores with a red light, and then expose the sample to a small amount of green light—so small that only a few of the fluorophores will activate at a time. An image of those scattered fluorophores can then be made and their positions determined to within nanometer accuracy. By repeating this process many times to locate the position of all fluorophores, the scientists can create a whole image.
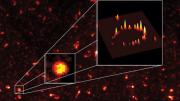
Zhuang’s team calls the method stochastic optical reconstruction microscopy, or STORM, and published a demonstration in a recent edition of Nature Methods. With this technique, “You’re going to get many of the advantages of optical imaging, but sharper and crisper by more than an order of magnitude [10 times],” she says. As an example, her team attached 20 to 40 fluorophores to a circular string of DNA and proteins and was able to resolve its ring-like structure, where conventional microscopy would have shown only a blob. Zhuang’s team is now hunting for other fluorophores that have the same ability to switch on and off, but in different colors.
The main drawback of the technique is the time it takes to gather multiple views and stitch them together. Creating an image can take a few minutes, making it best for viewing fixed structures. But Zhuang’s team hopes to improve it further, to be able to capture the movements of molecules in cells, which happen on the order of seconds to milliseconds. She says STORM is one of a few promising approaches that have the potential to turn high-resolution still images of the cell into live action. “We’d like to look at events in live cells with nanometer resolution, in real time,” she says. “That’s the hope.”
~Courtney Humphries
Xiaowei Zhuang e-mail address: zhuang@chemistry.harvard.edu
Website: https://zhuang.harvard.edu