Science is like fishing. Patience, perseverance, and skill are part of it. Luck also plays a large role. But finding the right location often makes all the difference. When Louis Agassiz, professor of zoology and geology and founder of Harvard's Museum of Comparative Zoology, first visited Woods Hole, Massachusetts, he must have realized the tiny backwater fishing port had potential. ("Hole," in the language of Cape Cod's early English settlers, referred to a passage between islands.) As a scientist obsessed with categorizing, comparing, and understanding animals—particularly fish—Agassiz noted that the local waters were packed with diverse specimens. Summers at the southwestern tip of Cape Cod were pleasant, and bustling Boston was comfortably distant—but not too distant.
 |
A yellow crinoid, a relative of sea stars, and a pink sea pen, possibly related to corals and anemones, inhabit the deep-sea floor. |
Photograph © 2002 Woods Hole Oceanographic Institution. Courtesy of the Galápagos Rift 2002 Expedition. |
In 1873, Agassiz began a utopian experiment in education on Penikese Island, a lonely chunk of windswept rock 12 miles south of Woods Hole. He envisioned a school where top teachers and students would study nature in its fundamental forms, free of distraction. Although he died later that year and the school quickly folded, Woods Hole's reputation, like that of a good fishing spot, had already spread. In 1888, a group of Agassiz disciples and colleagues from other universities founded the Marine Biological Laboratory (MBL), laying foundations that would one day transform the village into the world capital of marine research.
During the golden age of exploration—roughly the mid 1400s to the late 1700s—the goal was to navigate across oceans with all hands and cargoes intact. But around the time Agassiz waded into the muddy shallows off Woods Hole, science began to change the way humans understood the oceans themselves. Today, 130 years later, Woods Hole hums with scientific energy. Offices of the U.S. Geological Survey, the National Marine Fisheries Service, and the National Oceanic and Atmospheric Administration nestle along its narrow streets. And the MBL and Woods Hole Oceanographic Institution (WHOI) have made Woods Hole the maritime equivalent of Cape Canaveral—a forward base for exploration and an intellectual magnet for some of the brightest scientific minds on the planet.
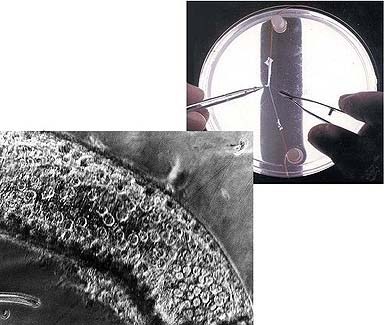 |
Above right: A scientist peels away the outer layers of an axon from the squid Loligo pealei. Left: Ring-shaped sacs called vesicles form after an axon has been injured and flow toward the wound, eventually sealing it in a process that may one day help scientists repair human nerve cells. |
Photographs courtesy of Harvey Fishman |
The ocean embraces up to 80 percent of all species on Earth, and comprises nearly as much of the planet's available living space. It regulates climate, absorbs greenhouse gases, and feeds humans.
For all that we benefit from the ocean, however, we know relatively little about it. Scientists estimate that thousands, perhaps millions, of species of microorganisms and larger creatures still live anonymously in its depths. The seafloor itself is a sinkhole of memory—where vast records of life, death, and cataclysmic change on earth during the last 100 million years or more may lie encased in mud and stone. Yet we have explored little more than 5 percent of the ocean bottom. In Woods Hole, the MBL and WHOI are dedicated to pushing deeper into this blue frontier in generally complementary fashion. MBL researchers explore biophysics, neurobiology, embryology, ecosystems, and marine resources using marine animals as models. WHOI scientists cover the disciplines of oceanography—physics, biology, chemistry, and geology—and also extend into marine engineering.
"Study nature, not books," Agassiz is said to have told his students. His fondness for hands-on learning still drives MBL scientists, says Gund professor of neuroscience John E. Dowling '57, Ph.D. '61, president of the MBL's corporation and a researcher at the lab since the late 1960s. "Scientists are still using the uniqueness of the organisms in the sea to study fundamental biological problems," Dowling points out. Over the years, the MBL has cultivated an atmosphere of freedom and flexibility and avoided formal ties to universities and government institutions while attracting scientists from around the world. Forty-eight Nobel laureates have worked, taught, or studied at the MBL, including H. Robert Horovitz, Ph.D. '74, who shared the prize for physiology and medicine in 2002 for investigations into how genes regulate organ development and apoptosis, or programmed cell death. What brings such talented people to the MBL? "Other people," says Dowling. "It's been sort of a who's who for world biologists since the beginning."
A year-round staff of about 275 runs daily operations and conducts research on the MBL's campus, which is loosely organized around specialized units such as the Josephine Bay Paul Center of Comparative Molecular Biology and its global infectious diseases program. But ever since its founding, the MBL has mostly been a summer research facility, hosting up to 1,400 researchers, students, and lecturers from the United States, Europe, Asia, and South America.
Researchers share labs and hunches. For students, the days are rigorous. Classes start around 8 a.m., says Dowling. Then, "at about 10:30 p.m., people end up at the Cap'n Kidd [a nearby bar], finishing their day with a beer or two. But only two—because they have to do it all again in the morning." Some students stay up even later, jostling for spots at the MBL's largest microscope.
Although most MBL investigators do basic research, many research tracks also have potential applications. For the last several years, the MBL has emphasized biomedical research; half a dozen faculty members and lecturers from Harvard Medical School show up for the summer. Some researchers seine the oceans like bio-prospectors, seeking useful microorganisms. Others study larger creatures. Horseshoe crabs, for example, with their lustrous compound eyes, have taught us about sight; pharmaceutical companies have harnessed chemicals in their blood to test medical equipment for harmful bacteria. Toadfish have blasted into space with shuttle crews for studies on balance and dizziness. Clams yield compounds that may be useful in cancer treatments. But nothing better illustrates research at the MBL than the long-finned squid, Loligo pealei.
 |
Above left: This Loligo pealei shows the intricate color patterning found in many cephalopods. The chromatophores, the freckle-like color-changing organs, can rapidly flash camouflaging patterns. The large movable eye helps the squid detect predators deep in murky waters. Cephalopod camouflage and vision are foci of the research done by Roger Hanlon of the Marine Biological Laboratory. |
Photograph by Jim Harrison |
With large luminous eyes, a graceful, rocket-shaped body, and skin that flashes up to 50 hypnotic patterns, the squid has been the model for many movie monsters. Otherworldly appearance aside, the cephalopods are amazing animals, hard-wired for speed, camouflage, and long-distance travel in a dark realm packed with predators. Loligo also possesses the largest axon—a kind of electric cable stretching between nerve cells—in the animal kingdom. This array of traits makes it the biological equivalent of a Swiss Army knife, useful for researchers investigating neuroscience, marine ecosystems, and the health of ocean fisheries.
"The magic of Woods Hole is the environment of having the facilities to do great research," says Harvey Fishman, a professor of physiology at the University of Texas Medical Branch at Galveston, a summer researcher at the MBL for nearly 40 years, and one of Loligo's biggest fans. Fishman has jumped research tracks several times during his career—from electrical engineering to cell biology to biophysics—always pursuing fresh questions and developing new skills to investigate his discoveries. The MBL has facilitated his progress. "All of the investigators who are here form a huge wealth of information," he says. "The mix is unsurpassed anywhere else in the world."
Fishman's latest leap has landed him on the cutting edge of neuroscience. His summer lab is a cinder-block cavern packed with computers, electrical equipment, sinks, and a tank for squid. Here he investigates Loligo's nervous system and explores nerve repair using the squid's giant axon. Like soup cans connected with string, neural cells communicate along the axon, firing electrical messages down the line. The axons are readily visible: they measure up to 10 inches long, and are about as thick as a pencil lead. Compared to minute mammalian nerves, they're fat as garden hoses.
About a decade ago, Fishman studied how ions traveled through the axon membrane and transmitted nerve impulses. He dissected the squid and tugged out axons with the cool dexterity of a fisherman tying flies, careful not to damage them because wounds shorted out their bio-circuitry. One day, he says, "I realized I had never, ever, looked at an axon that I had inadvertently injured. So I cut [one] and put it under the microscope, and lo and behold, all these magical things were happening."
Within an hour after the injury, vesicles—tiny fluid-filled sacs—had formed on the cell walls. The vesicles eventually broke away in large clumps and flowed toward the wound, plugging it and restoring electrical conductivity to the cell. "Nobody had observed this in 40 years of work on squid axons," Fishman says. "It's a very prolific process. My first impression was, 'How could this have been missed?'" More questions burst from his observations: What causes this process, called vesiculation? How does it restore electrical conductivity? Does this repair process occur in mammalian cells? Could it reverse nerve damage in humans?
Fishman has learned that calcium triggers the repair process. In small amounts, calcium ions regulate cellular changes and bind proteins. In large amounts, however, calcium kills, so when an injury allows high concentrations of calcium ions to seep into the axonal system, the cell responds by sealing the breach and flushing out the calcium overdose.
According to Fishman, research into nerve repair has generally been sidelined in favor of work on nerve regeneration. For years, experts thought nerve cells responded to injury by collapsing and fusing to seal a wound. "This notion probably comes from scientists who were studying mammalian nerves, which are very tiny," Fishman explains. "Without extreme magnification, it's hard to see what happens. But we've never seen [collapse and fuse] happen on any axon. It's vesicles that repair the cell membranes. That's a relatively unknown fact that we think is universal." Repair of the nervous system must begin with the repair of individual neurons, Fishman insists. Without these cells, the basic pathways of communication crumble.
Now Fishman is trying to understand how vesicles restore electrical conductivity to cells and to predict survival rates for injured nerve cells. So far he's found that axons punctured close to the cell body are less likely to live, because calcium destroys the cell body faster than vesiculation can plug the wound.
Using cultured cells, Fishman has also shown that similar neural patching occurs in mammals. He is reserved about his findings, but thinks they may one day help researchers launch a new era of nerve repair: some day, injured cells may be induced to fix themselves and reestablish neural pathways, thus preventing paralysis, organ failure, or brain damage. "The model system can sort of point the way, but you have to be very careful," he says. "We think it's really important [first] to understand cellular repair." For now, Fishman plans to let serendipity guide his work. "I feel I've been handed this incredible observation. People think they're going to manage science, but it inevitably doesn't go that way. Often out of disorganized observations comes a beautiful pattern that leads to understanding."
For MBL biologist Roger Hanlon, Loligo is a lot more interesting with its axons intact. Hanlon, an expert on cephalopods and director of the MBL's Marine Resources Center (MRC), has worked with squid for nearly 30 years. His friends tease him for spending his career studying bait. Hanlon shrugs it off—and ribs Fishman instead. "The standing joke, for the Harvey Fishmans of the world, is that a squid is just a support system for a giant axon," says Hanlon, grinning. "But here at the MRC our focus is on organismal integrative biology. We are looking at the whole organism: why they do the things they do and how they fit into the ecosystem."
This past winter, Hanlon collaborated with colleagues from the Woods Hole office of the National Marine Fisheries Service, the Massachusetts state fisheries service, and the local commercial fishing community to conduct a squid study that has complex implications for the future of marine-resource management. For years, Hanlon explains, experts believed Loligo populations comprised a single, homogenous genetic stock. Hanlon's group tested this theory, comparing genes of squid from five different near-shore locations along the East Coast, where the creatures spend the spring and summer spawning and feeding. They also hauled samples from several offshore locations where squid over-winter in deep canyons along the continental shelf.
The group found not one but five distinct genetic varieties. What's more, while squid from different genetic groups mingled during their winter retreat, the cephalopods separated out in the spring, forming genetically homogenous groups and returning to specific coastal areas. "We think they're going back to where they spawned," Hanlon says. "Scientifically, we may have the first demonstration of a marine invertebrate animal—a 'squishy,' as we say—that has the capability to migrate back to specific areas. We all know that salmon do this, whales can do it, turtles do it, and eels do it. How do squid do it? We don't know. It's amazing." According to Hanlon, the squid—already equipped with big brains, a dazzling array of sensory capabilities, and long- and short-term memories—may smell their way home.
These discoveries may reshape the rules that federal regulators have grafted onto marine ecosystems. Squid are big business: from calamari rings to axons, they support the fifth largest marine industry in the Northeast, generating up to $30 million per year. Their commercial appeal puts them at risk of being over-harvested; increasingly, fishermen pursue them in winter, catching them before they mate. The federal government has always managed the squid fishery as a single genetic stock. Now, Hanlon says, that plan is outdated.
In one of the MRC's classrooms, Hanlon draws his finger along a large bathymetric map of the East Coast and points to a spot south of Delaware Bay, where one squid group summers. "If this was one big homogenous genetic stock, then if you [fish] the hell out of [this] area, you're not going to reduce the gene pool," he says. But because the squid vary genetically, if one regional group is extirpated, it could take decades or longer for the animals to return, if they returned at all. Because higher predators such as marine mammals, fish, and birds depend on squid, the ecosystem—and human fishing fleets—would undoubtedly suffer in turn.
There is a national belief that the ocean is a bottomless pit of resources, Hanlon says. Indeed, the oceans provide nearly 100 million tons of food per year, about 20 percent of the world's dietary protein. Yet most adults don't understand the biological systems that bring them dinner. So far, Hanlon says, there has been only limited research into the genetic populations of cod or haddock. "It makes you rethink how the fisheries are being managed. Our trade deficit on seafood is huge. Our fisheries are collapsing. We have to [begin] managing the ocean with some biology and some modern conservation measures. It's one whole functioning ecosystem and the government hasn't really taken much notice of this."
In 1872, just a year before Agassiz opened his school on Penikese Island, Great Britain refitted a warship and launched it on an unprecedented mission: to haul as much scientific information as possible from the world's oceans. HMS Challenger's expedition showed that the oceans were far deeper and more mysterious, and heaving with more animal life, than anyone had ever imagined. Modern oceanography was born from this remarkable journey, and soon the same qualities that attracted biologists to Woods Hole began luring oceanographers.
In the late 1920s, the National Academy of Sciences began scouring the East Coast for a suitable spot on which to build an oceanographic institution. The Hole's harbor—sheltered, deep, and generally ice-free—made it an attractive haven for large ships. Not far off the eastern edge of Cape Cod, cold currents drop down from the Gulf of Maine while the warm Gulf Stream washes up from the south, creating a kind of two-lane highway for marine creatures and providing easy access to the ocean's enormous circulation system. These natural advantages, coupled with the village's already flourishing intellectual community, made Woods Hole the clear choice.
 |
Biologist Colleen M. Cavanaugh studies life around deep-sea rifts, such as the bacterial symbionts (below, from a transmission electron micrograph) that live within tissue of the hydrothermal vent tubeworm, Riftia pachyptila. The bacteria provide food for themselves and their hosts through chemosynthesis. |
Photograph by Jim Harrison |
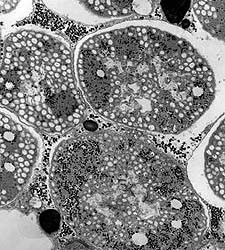 |
Photograph courtesy of Colleen Cavanaugh |
The Woods Hole Oceanographic Institution was founded in 1930 with a $2.5-million grant from the Rockefeller Foundation, on a lump of land purchased from the MBL. The institution's first three directors were Harvard graduates. Professor of zoology Henry Bryant Bigelow '01, Ph.D. '06, S.D. '46 (see "Vita," January-February 1999, page 46), steered the institution through its opening decade. He championed integrative studies of the ocean and urged oceanographers to do more than collect data. He also favored an experiential approach—insisting that his scientists go to sea at least once a year.
WHOI was fashioned in the image of the MBL, says James R. Luyten, Ph.D. '69, the current executive vice president and director of research. Like the MBL, WHOI remained a summer-only research facility until World War II. In 1940, Bigelow's protégé Columbus O. Iselin '26, A.M. '28, took over the directorship; at the outset of the war, he lent WHOI's facilities to the U.S. Navy and the institution was transformed into a year-round facility. Researchers made important discoveries about anti-fouling protection (which prevents drag and increases fuel efficiency), underwater explosives, and the interplay among temperature, salinity, and sound (which affected efforts to track submarines). When the war ended, Iselin's decision to maintain year-round operations shoved WHOI into the modern era.
Today, WHOI is the largest independent oceanographic organization in the world. It employs a staff of roughly 1,000, commands an annual budget of roughly $100 million, and operates three large oceanographic research vessels plus the DSV Alvin, the deepest-diving submersible in the U.S. scientific fleet. WHOI peers into the ocean through five intertwined departments (physics and engineering, ocean chemistry, physical oceanography, geosciences, and biology) and four internal institutes (dedicated to ocean exploration, deep ocean biology, coastal studies, and climate studies). But its specialists also communicate across disciplines, learning their colleagues' methods and vast vocabularies to better understand fundamental ocean processes.
Of all WHOI's research, none better illustrates the spirit of scientific exploration and interdisciplinary drive than the discovery in 1977 of hydrothermal vents in the crushing blackness at the bottom of the sea. Scientists skimming the ocean floor in Alvin first discovered vents more than 8,000 feet down off the Galápagos Islands. These undersea geysers, heated from below the earth's crust, have since been found elsewhere at similar depths, dotting the 46,000-mile Mid-Ocean Ridge system that rings the earth, it's been noted, like stitching on a baseball. Fluids heated by magma and loaded with hydrogen sulfide and metals such as copper, iron, and lead blast up through the vents at temperatures of up to 750 degrees Fahrenheit. As the fluids collide with near-freezing seawater, their loads precipitate out of solution, creating mineral and metal chimneys, some of them several stories tall.
But the sites are more than geologic wonders—they also throb with animal life under a constant rain of vent fluid. At some vents, tubeworms nearly seven feet tall grow in thick forests, towering above masses of giant mussels and clams. Eyeless shrimp, chalk-white crabs, and sluggish fish hunt and scavenge among their sessile neighbors. On average, a new species has been described every week and a half since the first vents were discovered. The vents also support huge populations of bacteria and of Archaea, single-celled organisms thought to be among the oldest life forms on Earth. Some evidence suggests a vast microbial carnival exists below vent sites, where hyperthermophilic microorganisms appear to thrive in cracks and pores in basalt rocks. Some researchers believe this "deep biosphere" could extend miles below the seafloor and harbor a biomass as extensive as that on the planet's surface.
WHOI scientists took leading roles in exploring the vents and their discoveries profoundly changed science. The existence of new life forms instantly smashed prevailing theories about deep-sea biology that had described the ocean floor as a great desert. The relatively young theory of plate tectonics gained support as vents became windows into the earth's interior. Chemists and oceanographers were also rewarded: the super-hot, mineral-rich vent plumes directly affect ocean temperature and chemical composition. Today, vent science, like oceanography, draws on dozens of scientific fields and sub-specialties and WHOI scientists continue making discoveries in the deep at an astounding rate. Nowhere else do so many disciplines join forces to study—and simply to visit—single ecosystems.
Sinking to the bottom of the ocean in Alvin can take as long as two hours. As the three-person sub descends, the blue ocean fades to impenetrable black. Crammed into Alvin's titanium belly, scientists probe the darkness with powerful lights and press their faces against the cold, damp glass of the tiny portholes. An average dive lasts between six and 10 hours. "It saps your energy," says Jeffrey professor of biology Colleen M. Cavanaugh, Ph.D. '85. "It's cramped, there's no bathroom. You keep putting on clothes to stay warm. But I never get tired of it." For Cavanaugh and many other scientists, the highly prized dives are scientific shopping sprees, fleeting chances to collect data from a world they can't reproduce in the lab. Cavanaugh's quarry is smallest of all: tiny, sulfur-loving bacteria.
Cavanaugh, who holds positions at both the MBL and WHOI, exemplifies the cross-pollination in Woods Hole. She studies prokaryotes—microorganisms without nuclei—investigating the symbiotic relationships they form with higher animals. Prokaryotes are nature's great alchemists, transmuting otherwise inedible substances into the carbon-based treasures that support life. Eukaryotes—cells with nuclei—depend on symbiotic prokaryotes for survival. More than 20 years ago, Cavanaugh rattled the emerging vent-science community by showing that these symbiotic relationships not only occurred at vents, but also made life possible.
In 1981, she was a Harvard graduate student listening to a visiting scientist puzzle over giant tubeworms hauled up from the Pacific. The worms lacked guts, mouths, and anuses. With such odd anatomies, the scientist wondered how they grew seven feet long in such extreme, essentially foodless, pitch-black environments. On the earth's surface, life depends on photosynthesis to provide the carbohydrates at the base of the food chain. What powered life on the seafloor? Cavanaugh found the answer in relationships that other biologists had missed, and offered a startling theory: what if tubeworms had developed symbiotic relationships with single-celled sulfur-oxidizing bacteria, which produced food not by photosynthesis but through chemosynthesis?
Cavanaugh reasoned that the bacteria lived in tubeworm tissue and used hydrogen sulfide present in vent fluid to convert carbon dioxide into carbohydrates, thus feeding both themselves and their hosts. With other researchers, she showed that in the cold, sunless sea, tubeworms became "effectively like plants on the seafloor." But instead of reaching up for the sun's rays, vent life plugged into the planet. "Bacteria can do anything,'' Cavanaugh says, grinning. "They keep the whole world going down there."
 |
Often called Woods Hole squid, Loligo pealei "seems almost custom-built for research," says physiologist Harvey Fishman. |
Photograph by Roger Hanlon |
 |
Like ruby lips, tubeworms extend their gill plumes and gather hydrogen sulfide from fluids at a vent on the East Pacific Rise. The sulfide is carried to symbiotic bacteria inside the worms (see image above). The large plumes at left belong to Riftia pachyptila; these giant worms may be the fastest-growing invertebrates on the planet. Nearby, a zoarcid fish cruises over a patch of smaller worms called Tevnia jerichonana. In the background, brachyuran crabs (Bythograea thermydron) scuttle through the worm forest. |
Photograph courtesy of Richard Lutz, Rutgers University, Stephen Low Productions, and Woods Hole Oceanographic Institution, with funding provided by the National Science Foundation. |
Last November, Cavanaugh returned to the floor of the Pacific aboard Alvin, stalking the symbionts that launched her career. She is interested in a new riddle: how do tubeworms and bacteria meet and agree to the intimate terms of symbiosis? Long ago, sets of free-living organisms apparently chose cooperation over digesting each other. Those relationships evolved into complex and essential biological processes. Human mothers, for instance, pass clutches of heirloom symbionts called mitochondria to their children; tubeworms apparently don't pass along their sulfur-oxidizing symbionts. "All of the data suggest that [the worms] get their symbionts from the environment," Cavanaugh says. "It doesn't make any sense to us. Are there free-living forms of the symbiont? How do the worms and the bacteria find each other? How has this come to some sort of détente?"
At the bottom of the sea, Cavanaugh believes, we can look back into the shadows of our own past. Some scientists have already posited that life may have evolved at vents, rather than in a shallow, sunlit goop at the surface, as the leading current theories suggest. Other researchers have argued that we never really left the oceans: that our ancestors' bodies evolved into the first spacesuits: cocoons of skin wired with veins, stuffed with bacteria, and swirling with fluids—the whole system designed to haul mini-oceans along onto land and keep them from drying out. Finding bacteria that exchange free lifestyles for symbiosis could provide a modern example of how the eukaryotic cell evolved from a microbial consortium, Cavanaugh explains. "We're trying to place the symbioses in the larger picture of [evolutionary] diversity. It's reorienting our thinking about how life evolved."
Hydrothermal vents are like islands of life scattered across the seafloor. They may also be ephemeral hotspots, blinking on and off to unknown geological rhythms. Despite the extreme conditions, the thriving life forms in vent communities are incredibly diverse. No one knows how the creatures evolved, how they colonize new sites, or how they maintain their populations.
WHOI staff biologist Timothy Shank is the deep-water equivalent of a backwoods tracker: he uses DNA to follow the evolutionary paths of vent organisms over time and map their modern migrations from vent to vent. As scientists first struggled to understand how vent animals evolved, some theorized that these were living fossils, perhaps originating in the Mesozoic or Paleozoic eras (from 65 million to 570 million years ago), and then evolving along isolated paths. Shank's research has shown that at least some vent species may actually be newcomers.
Vent shrimp don't look like the supermarket peel-and-eat variety. They're eyeless and highly specialized: some Atlantic species are equipped with photoreceptors on their backs that may "see" thermal radiation from vents, which glows at the infrared end of the spectrum. The shrimp seem like weird relics that have managed to lie low, really low, and escape the periodic extinctions that have ravaged the earth. But Shank has found that as a group, these shrimp are new kids on the block. By studying rates of genetic mutation, he has estimated conservatively that these shrimp diverged from their closest relatives to become a new species within only the last 20 million years.
This is remarkably recent, he says, compared to neighboring species like vent clams (about 60 million years old) and giant tubeworms (roughly 100 million). His findings suggest that vent communities have not necessarily been cut off from the rest of the ocean for eons. Like shrimp, other species may prove to be recent immigrants, perhaps arriving in explosive evolutionary spurts, hopping from shallow areas to progressively deeper regions, filling vacant ecological niches at vents.
On a smaller scale, Shank is fine-tuning techniques of genetic fingerprinting that will allow him to match vent animals with their closest relatives. This is crucial, he says, in understanding how larvae travel through the undersea wilderness. Early studies linked certain vent communities genetically, proving that the animals successfully launched their young into the deep and colonized new sites. But unknown forces also shape vent populations. Researchers found that in the Atlantic, for example, great clouds of translucent shrimp swarm over vents like ghosts, and several different species dwell at the vents. In the Pacific, however, each vent harbors a separate shrimp species. Tubeworms offer another mystery: they've been found only in the Pacific. How do some animals radiate across broad expanses of seafloor, while others, seemingly, are corralled into geographic pens?
"We know that [vent invertebrates] cast large numbers of larvae into the water column," Shank says. "[But] how they zero in on a vent as target, no one knows. What is fascinating is that whenever a new vent opens up, certain animals begin to colonize it almost immediately." So far, he and others believe that larvae travel in currents, and that chemicals in vent fluids may act like beacons. Shank hopes to obtain discrete DNA measurements of animals within specific sites. When combined with oceanographic data (current directions and speeds, chemistry, temperature and pressure), the measurements, he believes, will help him infer migration patterns and decipher how larvae home in on vents.
If currents are the highways on which larvae travel, however, then geologic features may be the detours, roadblocks, and speed bumps that foil them. Shank thinks he's identified some of the barriers that account for differences in vent fauna. Oceanic ridges almost certainly play a part by redirecting currents. Major fracture zones along tectonic plates also may play a role, he explains, somehow rebuffing or perhaps trapping packets of larvae. Somewhere near the Equator, there's yet another filter, he believes. There, a combination of currents and a shallow, ocean-bottom ridge or sill may trap larvae in the southern Atlantic while keeping other migrants out. The resulting stew of organisms could be phenomenally bizarre, shaped by isolation and inbreeding.
 |
Above and below: Eyeless shrimp (Rimicaris exoculata) feed at a vent on the Mid-Atlantic Ridge. Juvenile Rimicaris are reddish; in adulthood, they become translucent or pale gray. Scientists believe the iridescent V-shape behind their heads may be a photoreceptor, possibly capable of "seeing" geothermal energy radiating from the vents. |
 |
Photographs courtesy of Richard Lutz, Rutgers University, Stephen Low Productions, and Woods Hole Oceanographic Institution, with funding provided by the National Science Foundation. |
In April, Shank shipped out to the Mid-Atlantic Ridge aboard WHOI's research vessel Atlantis to investigate Lost City, a new kind of vent site that sits in relatively shallow water (about 2,500 feet). The geology and chemistry at Lost City differ from most other known vents. The fluids are cooler and loaded with methane instead of hydrogen sulfide. Previous investigators also reported fewer animals at the site—but there may be new types of shrimp.
For Shank, the discovery of such a biologically and chemically different system underscores the importance of vent research as a whole, work that is no less crucial, he says, than expeditions into space. "In the next 10 years, studies of novel, extreme environments are going to change how we think about how animals interact with their environment, and there's going be an impact...on basic evolutionary science," he says. So far, "We know a little about the Indian Ocean, and in the Arctic and South Atlantic we know nothing. We're still in the Lewis and Clark phase. We have no idea what's on the other side of the mountain."
For more than a century, Woods Hole has been a national window on the oceans. From village docks, scientists have sent expeditions to plumb the deepest parts of the sea. In local labs, they have probed the miniature universes of cells and genes. Now, with its troves of data, Woods Hole may provide an intellectual launch pad for new types of research in space. Extreme temperatures and differences in pressure and unexplored territories await oceanographers in their fragile, deep-diving capsules. No other environment on Earth demands so much of its inhabitants or so reminds scientists of conditions in space.
When scientists dream of life beyond Earth, Europa—Jupiter's fourth-largest satellite—is the moon that spins through their visions. Slightly smaller than Earth's moon, it is at once the most exciting and puzzling body in the solar system because conditions there may support life. For scientists searching out extraterrestrial organisms, the journey may begin in the deep sea, where possible analogues for life in space exist at hydrothermal vents.
Life as we know it demands water, energy, and nutrients. Europa may have all three in abundance. Data gathered over the last several years from the Galileo spacecraft show that a crust of water ice, estimated at 3 to 12 miles thick, encases Europa. This outer layer may protect an ocean up to 30 miles deep. The best evidence yet for liquid water is magnetic. Magnetic readings of Europa suggest that some kind of electrically conductive layer encompasses the moon near its surface. Solid ice is a poor conductor, but seawater, according to one scientist at the National Aeronautics and Space Administration, "is about right."
 |
Biologist Timothy Shank aboard Alvin, the deepest-diving submersible in the U.S. scientific fleet. |
Photograph courtesy of Richard Lutz, Rutgers University, Stephen Low Productions, and Woods Hole Oceanographic Institution, with funding provided by the National Science Foundation. |
Europa also appears to be composed of rocks similar to those found on Earth, and spectral analysis of strange brownish streaks along its surface suggests the presence of sulfate salts. If vents do exist, the chemical and mineral mix may be just right to provide nutrients for life. Assuming that Europa has all the ingredients on life's list, what sorts of organisms might be found there? Most likely, say Colleen Cavanaugh and many of her colleagues, they'll be microorganisms. On Earth, such creatures can live just about anywhere, from jet-plane fuel tanks to Antarctic wastelands. Archaea, the ancient form of single-celled organism found at vents, thrive in the ultra-hot vent environment; even hardier microorganisms have been found living inside the earth's crust.
Recently NASA decided to proceed with plans for the Jupiter Icy Moons Orbiter (JIMO), a nuclear-electric propelled spacecraft, that would circle Europa and its frigid companions, Callisto and Ganymede. Scientists hope to establish a kind of geodetic and oceanographic platform from which to test theories about the age of Europa's icy crust, the depth of its liquid stores, and the evolution of the moon itself. With their vast knowledge of marine life and ocean systems, MBL and WHOI researchers are well positioned for a search for extraterrestrial life on potentially watery moons. From the blue frontier to the vastness of space, Woods Hole is proof that sometimes science just needs a good spot to start.
Freelance writer Neil Shea is also a reporter at the Providence Journal.