Today’s high-powered light microscopes bear little resemblance to the iconic instruments of high-school biology labs. This revolution began in the 1950s with the development of confocal microscopes. Rather than flooding an object with light and viewing its surface, confocal microscopes use a highly focused laser beam to quickly scan an object in one plane. By collecting information only from a single “slice” of a sample at a time, confocal microscopes reduce the effect of surrounding visual noise. Scientists can gather many of these slices at different depths to give a three-dimensional picture.
Equally important to the new wave of microscopy are the fluorophores that tag molecules and cells with different colors. A green fluorescent protein (GFP) was first introduced in labs in the early 1990s. Since then, fluorescence imaging has become a ubiquitous technology in biology, and species from fish to pigs are genetically engineered with cells and proteins that glow green. In addition to genetically engineering fluorescence into animals, scientists can attach fluorescent molecules to “probes” that stick to a protein of interest. Ralph Weissleder says that scientists are constantly uncovering new probes that emit light in a rainbow of colors. Many come from newly-discovered species in the ocean, while others are synthesized in the lab.
Most fluorescent molecules require blue light to excite them, but blue light is easily scattered by tissue and can damage cells. Multiphoton microscopes get around the problem with a trick: when two photons from lower-energy light hit the same fluorophore nearly simultaneously, they cause it to fluoresce as it would in response to a single beam of light that had double the energy. These microscopes therefore use red or near infrared light in brief, intense pulses focused on a small point at a time. The light is not as damaging to tissues and can travel deeper—about 500 hundred microns deep, which is just the width of five human hairs but represents dozens of layers of cells into a tissue.
The third factor that has made dynamic, live imaging possible is computing power. The colorful images produced by these techniques result from collecting and interpreting large amounts of data, which was not possible until recently.
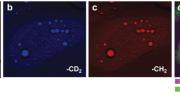
Scientists are also working on new imaging techniques that do not rely on fluorescence, since the use of fluorescent molecules in humans raises logistical and safety concerns (no human will ever be genetically engineered to fluoresce, and fluorescent probes are making their way into the clinic but may not be practical for many applications). Sunney Xie’s group, for example, has developed a technique called CARS (coherent anti-Stokes Raman scattering) microscopy that can image specific molecules in living cells and organisms based on their ability to vibrate at unique frequencies. CARS has so far been useful for visualizing the distribution of drugs in tissues and fats in cells—a recent study, for instance, showed in vibrant color how the presence of omega-3 fatty acids in liver cells helps them to break down fats. Ideally, different imaging technologies can be used in parallel to gather many kinds of information at once.