A thimbleful of your blood. To Robert Gerszten, that’s like a window on your well-being. In some cases, it may even let him see into the future of your health. Gerszten and his colleague Greg Lewis work at the leading edge of an emerging new field—metabolomics—that promises powerful insights into the mechanisms of human health and disease. They study blood metabolites: circulating small molecules, such as amino acids, lipids (fats), nucleotides, and carbohydrates, that are involved in metabolism. Metabolites not only reveal much about your current health—how well you burn fats or how deep you can dig when exerting yourself physically—but can provide hints of what’s to come. Research on the major metabolic killers—diabetes, kidney disease, and heart disease—reveals critical signs of systemic dysfunction at the molecular level years before clinical symptoms appear.
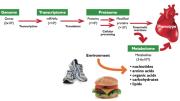
In their efforts to understand the human body and cure disease, scientists thought knowing all the 20,000 to 25,000 genes in the genome would lead to answers. But the genome encodes—as a rough approximation—more than a million proteins, each with a special function. Even more frustrating, changes to those proteins (from bonding with lipids, carbohydrates, and so on) lead to more than 10 million functionally distinct modified proteins. Compared to unraveling the effects and associations with illness of these numerous entities, metabolomics has a decisive edge: there are only, at current best guess, about 3,000 to 6,000 metabolites of interest.
Metabolomics, so named because it describes the metabolic products of the human genome, has existed—at least as a modern scientific concept—since Linus Pauling proposed in the 1950s that one could study breath condensates in order to capture human physiology. But the field has taken off only recently, explains Lewis, a cardiologist at Massachusetts General Hospital (MGH) and an assistant professor of medicine at Harvard Medical School (HMS). “If you look back to 2002,” he says, “there were very few cited papers in the world literature, and many of those [looked] only at the metabolites contained in a single cell. Even as recently as 2005, there were very few human studies.” But a steep increase in the number of published papers demonstrates how the field has grown exponentially since. Improvements in technology and lessons from genomics, proteomics, and transcriptomics—other broad, data-intensive disciplines of biological science and engineering—have fueled the pace of discovery.
Unlike genomic and proteomic strategies for probing human disease, metabolomics may make an almost immediate clinical impact by allowing doctors to deliver personalized medicine, based on metabolic profiling of patients. In an astonishing breakthrough published in Nature Medicine in March, for example (see “The Fingerprints of Diabetes…,” page 29), Gerszten and colleagues describe markers that identify people likely to develop diabetes more than a decade before any sign of the disease itself appears.
Gerszten, the director of clinical and translational medicine in MGH’s cardiology division and an associate professor of medicine, explains: “A gene makes transcripts, transcripts make proteins, and proteins (or enzymes) make metabolites.” Many steps separate a gene from its ultimate expression, perhaps as a disease, meaning that someone with a gene variant linked to heart disease may never become sick. But because metabolites are “downstream of genetic variation, transcriptional changes, and post-translational modifications of proteins,” Gerszten says, they capture what is actually happening in the body: “They are the most proximal reporters of any disease status or phenotype.”
They also capture the environment. “If you eat some noxious metabolite in your Big Mac,” your blood will reflect that, he says. Genes won’t.
For these reasons, Gerszten believes the metabolome is “equally, if not more, important than the human genome” for capturing “the fingerprint of human disease.” “Say you go to the doctor and have a blood test,” he continues. “What do we capture in terms of metabolism? Not a ton”: cholesterol, triglycerides, and glucose. He points to a densely lined, heavily annotated chart on his computer that looks as if someone has been twisting strands of spaghetti into patterns with a fork (see illustration above). “That’s every known metabolic pathway,” he explains. “Ideally, that is what we would be tapping if we had the tools.”
The Exercise Effect
Last year, Gerszten and Lewis ran an experiment to examine the effects of just 10 minutes of exercise on about 200 common metabolites. The pair had asked a group of patients that included healthy people—the “worried well”—to run for approximately 10 minutes on a treadmill. Blood samples were taken before exercise, at peak exercise as the test ended, and again an hour later.
Both men knew that exercise confers many health benefits, and Lewis, a former Olympic rower, has a background in exercise physiology—the study of the physical changes that take place in the body in response to exercise. Fit individuals, for example, have greater blood volume, more efficient hearts, and a better ability to extract oxygen from the blood for the use of working muscles; even their mitochondria (the energy-producing organelles within cells) are more abundant. Finally, after adjusting for age, peak exercise capacity is the most powerful known predictor of the risk of death among healthy people and those with cardiovascular disease. Even among those with cardiovascular disease, Gerszten notes, those individuals who nevertheless can exercise intensely on a treadmill have the same risk of sudden death as “the guy who has never had a diagnosis of heart disease.” The challenge is to understand why exercise counteracts disease.
Despite the brevity of their treadmill challenge, Gerszten and Lewis discovered that profound changes took place in the biochemical profiles of their patients’ blood. Glycerol, a marker of the body’s ability to burn fat, was up. Glucose-6-phosphate, an indicator of the use of energy stored as glycogen in the liver and muscles, increased. So did pantothenate, a modulator of fatty acid oxidation, and allantoin, a marker of oxidative stress. The human body turns on the systems of energy utilization and the mechanisms for dealing with the potentially harmful by-products of oxidative processes (free radicals) very quickly.
The study not only validated the usefulness of circulating metabolites as a snapshot of metabolism, it demonstrated that metabolic changes can be persistent. “Take-away message number one,” says Gerszten, “is that the blood is a beautiful place to sample.” That had not been entirely obvious, he says, because the by-products of many metabolic processes were thought to be retained within cells. But with “exquisitely sensitive new techniques” being pioneered at the Broad Institute of MIT and Harvard, where Gerszten helped launch the metabolomics program, “we can now pick them up outside cells.”
Take-away number two is that “an hour after exercise, more things had changed than at peak exercise”—as though exercise had initiated a cascade of signals throughout the body. After 10 minutes of exercise, heart rate and blood pressure return to normal in five or 10 minutes, but the metabolic changes persist. The study supports epidemiological claims that three spells of exercise a day, each of as little as eight to 10 minutes’ duration, are enough to confer health benefits. “These are the quintessential metabolic data,” Gerszten says, “suggesting that even short bouts have unanticipated prolonged effects.”
But the study also raised more questions than it answered. Fit participants, defined as being in the upper half of the sample, burned fat twice as well as the less fit. “There was more than a 100 percent increase in glycerol in more fit individuals,” reports Lewis, “compared to less than a 50 percent increase in the less fit.” When they extended the study to include runners in the Boston Marathon, they discovered that participants’ post-race lipolysis—the rate their bodies burned fat—was 11 times the rate of the least fit group. The marathoners had run for three and a half hours, but Gerszten believes that even if they had run only for 10 minutes, they would still have been in the highest quartile.
The researchers also noticed that levels of niacinamide, a compound that can help prevent or alleviate the symptoms of diabetes, rose most in the leanest individuals. “Now all of sudden we have these markers that distinguish what is happening in the more fit versus the less fit,” says Gerszten. “The million-dollar question is: Is that because of training, genetics, or diet? Those answers are what we are working toward.”
What they do know is that the metabolites that increase with exercise are not just markers of health—they actually promote it. They found that sprinkling muscle-cell specimens in a petri dish with a cocktail of the six metabolites that had multiplied the most activated a gene program involved in energy balance. They are now testing the dietary effect of this combination by feeding metabolites to laboratory mice.
They have also devised a human experiment to discover where the metabolites are created and how they are distributed during exercise. All organs and tissues in the body generate metabolites, but “our working hypothesis is that exercising limbs are sending global signals to the body to do certain things,” Gerszten says. “We now have to prove that.” As a first test of this theory, the researchers catheterized 10 individuals pedaling a stationary bike, in order to sample blood simultaneously from two different arteries: one tied to the general circulation and one more closely tied to the working muscles in the legs. This allowed them to compare the concentration of metabolites from the two locations and to discover that they are enriched in blood from the legs, probably generated during muscle contractions.
Their findings in the exercise test are “just an initial description,” Gerszten emphasizes. They have no idea why these metabolites cause beneficial changes, or why they work only in combination, or even how long the effects of these metabolic changes last.
Further research will answer these questions, but Lewis already sees the potential for using metabolic signatures to help his heart patients track their health progress as they follow a prescribed regimen of weekly physical activity. He has embarked on studies of the effects of exercise training among a broad spectrum of individuals, including employees of the hospital, seeking to define normal metabolic signatures by age, sex, ethnicity, and fitness level. “It may be that people have different responses to training,” he explains, and that might affect their metabolic profiles differently. Some people quickly lose weight when they exercise, for example, while others cannot.
But even though metabolic signatures may vary, Lewis suspects that the chronically overweight can still become fit, and that detectable metabolic changes occur to some extent in everyone. One metric commonly used to measure whether individuals are benefiting from exercise, he points out, is whether they lose weight—and “some people get discouraged if their weight doesn’t change after six to eight weeks of a training program, and they quit. If you could show people tangible evidence—‘These six metabolites that predict the future onset of cardiovascular disease changed 35 percent by virtue of doing this exercise’—that is potentially powerful” as a motivational tool.
Manipulating levels of metabolites to improve health—to use them as drugs— may also be possible, Lewis says. “These are naturally occurring small molecules already in your body, not potentially caustic drugs that may have side effects we will find out about only 20 years after people are exposed to them.” In fact, he has a bottle of one particular metabolite, glutamine, on a windowsill in his office. A white powder that can be purchased at any health-food store, it is metabolically active if ingested, and has the potential to regulate glucose tolerance.“Some people,” he says, “have this angry response to the idea of creating a pill that could substitute for exercise: ‘How could you?!’ they ask. Nobody is interested in dissuading people from exercise. But if we can have a better understanding of what is happening in response to exercise, then we can truly embark on a pathway of individualized programs that will optimize the health status of each patient. I think it is really hard to argue with the merits of doing that, particularly for people with diseases that preclude them from exercising.”
The Future of a Field
Gerszten and Lewis’s exercise research is a “perturbational study,” in which participants serve as their own controls. They have conducted such studies before and after weight loss, before and after the administration of drugs (to track drug levels and observe the metabolic changes they cause), and even before and after planned heart attacks (used to reduce the size of enlarged hearts), in which they identified metabolic markers associated with the death of cardiac muscle. Ultimately, they hope to develop metabolic profiles for patients that will allow them to identify physiological factors that limit performance. Is it the skeletal muscles? The heart? The lungs? Knowing the metabolites involved might lead to interventions that range from something as simple as putting supplements in a drink (Gerszten and Lewis are exploring the potential of so-called nutraceuticals, and have been in touch with a sports drink manufacturer) to new ways of investigating pathways involved in disease, to gain better understanding and, eventually, cures. And because some dietary metabolites appear to be harmful, Gerszten is feeding them to mice to learn whether they lead to atherosclerosis and diabetes.
Researchers today recognize that the relative contributions of genetics, behavior, and environment to maintaining health, on the one hand, and triggering metabolic diseases (even including some cancers), on the other—remain unclear. “Part of me wants to believe that there is some precise molecular pathway and an enzyme that we are going to identify and make a drug for,” Gerszten admits. “But then the other side of me slaps my head and says, ‘Wait a minute. Let’s hope it is not a molecular pathway, but something where we can intervene in a much easier way.” A silver bullet would be nice, he agrees, but most likely, the metabolic disorders will turn out to be complex. “Watching what you eat can help a hell of a lot,” he says, “but for some people, there will be a genetic component as well.” Metabolomics, he predicts, will help to provide those answers.